AMERICAN SOCIETY OF OCULARISTS |
Surgical Procedures
1) Loss Of An Eye
Losing an eye to trauma, tumor, or end stage ocular disease such as glaucoma, or diabetes can be devastating at any age. It may have a major impact on one’s self-image, self-confidence, and self-esteem, not to mention the adjustment required in adapting to monocular (single-eye) vision. There may even be some job restrictions that apply for one-eyed patients (e.g., commercial drivers, airline pilots, policemen, firefighters, etc.). Numerous patients have been fitted with prosthetic eyes who are currently employed in the professions mentioned above. Monocular patients should try to return to their work if possible and lead as normal a life as possible. Although there is some loss of depth perception and peripheral vision, they are not as “handicapped” as many believe.
2) Primary Socket Procedures
a) Enucleation Surgery - Enucleation refers to removal of the globe (eyeball). Surgery may be done under local stand-by anaesthesia (twilight anaesthesia) or general anaesthesia (patient asleep). It is commonly done either as an outpatient or overnight stay patient. The surgical procedure generally takes about 1 hour. While under anaesthesia, the lids are held open and the external coats of the eye (conjunctiva and Tenons) are trimmed away from the eyeball. The extraocular muscles (responsible for moving the eye into different fields of gaze) are also trimmed away from the eye surface. Lastly the optic nerve is cut and the entire eyeball is removed. Bleeding is controlled by gentle cauterization.
 |
| Figure 1: Examples of orbital implants over the past 20 years (right side of photo), and artificial eyes (left side of photo). |
When someone loses an eye, two components are needed: an orbital implant to maintain the volume of the eye socket and an artificial eye or prosthesis.
Thus, following removal of the eye, an orbital implant is put into the socket and the tissues are closed over top of it. The extraocular muscles can be hooked up to the implant surface to help keep the implant from migrating and to help with socket movement. Initially, a temporary prosthetic conformer made of clear plastic is put in place over the orbital implant. This conformer maintains the tissue space behind the eyelids where the prosthetic eye will eventually sit. In approximately 6-8 weeks, the conformer is removed and a custom made artificial eye (prosthesis) is made for the socket. Eye drops or ointment are required during the first few weeks post-op. Pain is something patients worry about routinely with enucleation surgery. There may be some discomfort post-operatively in the first few days but how much is variable, as everybody has a different pain threshold. What might be a lot of pain for one person may only be mild to moderate in another. Whatever pain is present (usually not severe) it subsides in the first 3 to 5 days. Pain killers are routinely prescribed after enucleation surgery along with antibiotics.
b) Evisceration Surgery - Evisceration surgery refers to the removal of the inside contents of the eye (cornea, iris, lens, vitreous, and retina). The white shell of the eye (sclera) is left in place. The extraocular muscles are left attached to the eye surface and the optic nerve is not cut. Once the contents of the eye are removed, an implant is placed into the scleral shell. The sclera, Tenons, and conjunctiva are then closed over the implant and a temporary (clear plastic) conformer is put in place. The real artificial eye (prosthesis) is made in about 6-8 weeks.
Evisceration, like enucleation, can be done under local stand-by (twilight anaesthesia) or general anaesthesia and may be performed as an outpatient or as an overnight stay visit. Like enucleation, pain is variable and dependant upon one's pain threshold. Generally, there is some pain but usually it is not severe. It settles in the first 3 to 5 days.
 |
| Figure 2a: Blind, painful, disfigured right eye with obvious scar. |
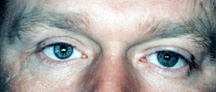 |
| Figure 2b: Following evisceration surgery and revision of scar. |
Which is best: Enucleation or Evisceration? Evisceration is quicker and simpler to do. There is less anatomical disruption to the eye socket with evisceration and the end results (appearance, movement) are superior in most cases then with enucleation. However, there are some situations where one simply cannot have an evisceration. In any eye that has a tumor (e.g., melanoma) or suspected tumor, an enucleation is the only option. If the diseased eye has end stage glaucoma, end stage diabetes, is a post trauma eye, has a known history without suspicion of a tumor, an evisceration is suitable and becoming increasingly common because of the ease of surgery and the excellent cosmetic results (Figure 2a, b).
c) Secondary Orbital Implant Surgery - In some individuals who previously had an enucleation procedure years ago, the orbital implant may have shifted out of position, become exposed, infected, or is simply too small. In this situation a secondary surgery can be performed to remove the first implant (secondary orbital implant surgery). Secondary orbital implant surgery can be more challenging than enucleation or evisceration especially if the surgeon tries to localize and reconnect the extraocular muscles. It is therefore usually done under general anaesthesia as an overnight stay patient. The post-operative healing is similar to enucleation and evisceration surgery.
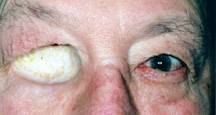 |
| Figure 3a: Exenteration socket (a skin graft that appears much lighter than the surrounding skin is lining the eye socket cavity). |
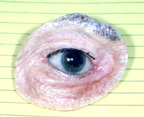 |
| Figure 3b: Oculo-facial prosthesis |
 |
| Figure 3c: The oculo-facial prosthesis is sitting in the eye socket. The glasses camoflauge the prosthesis |
d) Exenteration Surgery - Exenteration is a more radical procedure than enucleation, evisceration or secondary orbital implantation. It involves removal of all the tissues within the entire eye socket. The conjunctiva, globe, extraocular muscles and orbital fat are all taken out. In these situations there is no place to put an orbital implant or artificial eye as described above. An oculo-facial prosthesis can be made however, to cover the eye socket opening. Although it does not move, it looks much better than an empty eye socket (Figure 3a, b,c).
3) Orbital Implants
Prior to 1885 orbital implants were not used. The eye was removed by enucleation or evisceration and the socket was left to heal in its own. The result was an unsightly sunken depression of the eyelids into the eye socket. The use of an orbital implant was a major breakthrough in anophthalmic socket surgery. The implant improved postoperative cosmesis by filling orbital volume and also reducing the chance of socket contractions due to scar tissue formation. Over the last 100 years a variety of materials have been used for the orbital implant including: gold, silver, cartilage, bone, fat, cork, sponge, rubber, paraffin, wool, asbestos, as well as a variety of others in an attempt to find the most biocompatible implant (Figure 1).
A variety of shapes and sizes have also been tried in an attempt to promote some motility to the socket. In 1985 a new concept in eye socket implants began to evolve when a researcher (Dr. Arthur Perry, San Diego, CA) began to study sea coral as an ocular implant. Through a patented hydrothermal process the calcium carbonate component of sea coral was turned into calcium phosphate and a substance known as hydroxyapatite was made. Hydroxyapatite has the same chemical structure and porous structure as human bony tissue (Figure 4a).
 |
| Figure 4a: Hydroxyapatite orbital implant |
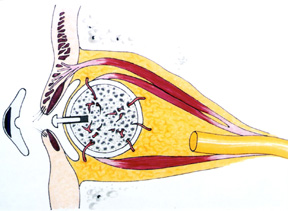 |
| Figure 4b: Drawing of hydroxyapatite implant with muscles attached and a peg in position |
 |
| Figure 4c: Synthetic hydroxyapatite implant. |
 |
| Figure 4d: Spherical porous polyethylene. |
The implant material (corralline hydroxyapatite) is biocompatible, non-toxic and non-allergenic. The body’s tissue recognizes the material as similar and because of the porous nature, tissue will grow into it. The implant becomes more fixed and therefore resists migration. The implant allows attachment of the extraocular muscles which in turn leads to improved orbital implant motility. The orbital implant can also be directly attached to the prosthesis through a peg, protruding from the implant (Figure 4b) allowing a wide range of prosthetic movement as well as the darting eye movements commonly seen when people are engaged in conversation. The increased range and fine darting movements allow a more life-like quality to the prosthetic eye.
The hydroxyapatite implant, also known as the Bio-Eye™ (integrated Orbital implants, San Diego) is called a “porous implant.” Since its introduction, a number of other “porous implants” have been introduced. One such implant (popularized by Dr. D.R. Jordan - University of Ottawa Eye Institute, Ottawa, Ontario, Canada) is a synthetic variety of hydroxyapatite (FCI3 HA implant) that has similar qualities to the original Bio-Eye™ but less expensive (Figure 4c). It is available in Canada and other parts of the world but due to patent restrictions is unavailable in the United States. Another synthetic porous implant that has become increasingly popular is a type of porous plastic known as Porous Polyethylene (Medpor™ - Porex Surgical Inc, Cooledge Park, GA). This material is a synthetic man-made material that previously has been used in a wide range of cranio-facial reconstructive procedures and facial fractures, with few problems. Its properties demonstrate high tensile strength, malleability, biocompatibility and fibrovascular ingrowth. The porous polyethylene orbital implants are also cheaper than the original Bio-Eye™, and are available in spherical, egg, conical or mounded shapes (Figure 4d).
Aluminum oxide (Al2O3) another man-made biomaterial that has been in use for more than 30 years as an implant in orthopedics and dentistry has also been studied extensively (Dr. D.R. Jordan - University of Ottawa Eye Institute, Ottawa, Ontario, Canada) and is emerging as an orbital implant in Canada, Europe and several other parts of the world. The aluminum oxide implant, also known as the Bioceramic Implant (FCI, Issy-Les-Moulineaux, Cedex, France) looks identical to the Bio-Eye™ hydroxyapatite with multiple interconnected pores. Like the synthetic HA and porous polyethylene, it is less expensive than the Bio-Eye™. There is evidence that human osteoblasts and fibroblasts appear to grow better on aluminum oxide than hydroxyapatite, suggesting it may be more biocompatible than hydroxyapatite and better tolerated in the eye socket.1,2,3
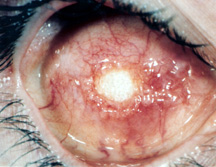 |
| Figure 5a: Exposure of a hydroxyapatite implant. |
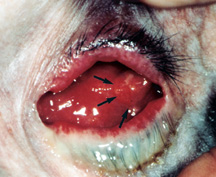 |
| Figure 5b: Orbital implant infection - the eye socket is very inflamed, there is lots of discharge and a recurrent pyogenic granuloma (black arrows). |
4) Orbital Implant Complications
Complications associated with hydroxyapatite that also apply to other porous orbital implants have gradually come to light since the introduction and widespread use of hydroxyapatite in the early 1990’s. Reported complications include: implant exposure, conjunctival thinning, discharge, pyogenic granuloma formation (excess healing tissue formation), and rarely persistent pain or discomfort. The complication discussed most often is implant exposure with exposure rates ranging from 0 to 22% (Figure 5a).
Predisposing factors to exposure include: wound closure under tension, inadequate or poor wound closure technique, infection, mechanical or inflammatory irritation from the speculated surface of the HA implant and delayed ingrowth of fibrovascular tissue with subsequent tissue breakdown.
The most feared complication of porous orbital implants is infection within the implant. Porous orbital implants have multiple interconnected pores that fill with fibrovascular tissue over the first 6-12 months which theoretically should help resist infection. Prior to this time implant exposure can predispose the implant to entry of bacterial contamination and implant infection. Once an implant infection does occur, it may not be easy to recognize or treat. Hallmarks of implant infection are, recurrent discharge resistant to multiple drops, implant discomfort (to touch), and recurrent pyogenic granuloma (excess healing tissue) on the surface of the implant (Figure 5b).
Implant infection does not respond to oral, intravenous and/or topical antibiotics and generally the implant has to be removed. Removal of an infected implant requires general anaesthesia and is traumatic to the eye socket as the implant has been partially integrated with the socket tissue. Following removal of the porous orbital implant it is not advisable to have another porous implant until a sufficient length of time (at least 6 months) has past to ensure tissue healing. A plastic nonporous sphere can be put in to maintain volume while waiting out the 6-month period and contemplating another porous implant.
5) Artificial Eye Motility: Implant Pegging Procedures
a) To peg or not to peg? One of the many advantages of porous implants (hydroxyapatite, porous polyethylene, aluminum oxide) is the ability to integrate them with the overlying artificial eye through a peg system. By coupling the orbital implant to the artificial eye a wide range of prosthetic eye movements as well as darting eye movements commonly seen in conversational speech can occur. These movements impart a more life-like quality to the prosthetic eye. To peg or not to peg, is up to the surgeon and patient. They are certainly not for everyone and with all due respect, not all eye socket surgeons are equally skilled at putting them in. Before considering a peg the implant has to be fully vascularized (minimum of 6 months, in some this may take a year or more) and, the socket has to be a healthy one. Patients with diabetes, previous radiation, systemic disease such as Systemic Lupus Erythematosis (SLE) or, individuals on medications such as steroids, are not good candidates for pegging, because their socket tissue simply does not have good blood flow.
Pegs are not fool proof and do have their own inherent set of problems above and beyond those of the implant. A meticulous peg placement technique is required to obtain excellent results; a fact not appreciated by many. The pegs must be central and straight. They must also be flush with the implant with no exposed portion to ensure a good result. Once pegged, the individuals do require regular follow up initially to be sure the peg is sitting well. With time the follow-ups can decrease (yearly) if all is well. The most worrisome problem associated with pegging is introduction of infection to the implant, requiring implant removal.
Other potential peg problems include discharge, pyogenic granuloma (excess healing tissue) around the peg, peg falling out, poor transfer of movement, clicking, conjunctival overgrowth, poor fitting or loose sleeve, part of sleeve shaft visible, peg drilled on an angle, peg drilled of center, HA visible around peg hole, and excess movement of peg.
 |
| Figure 6a: Polycarbonate peg system - original standard peg is to the right (black arrow); the peg and sleeve system to the left with screwdriver for sleeve below. |
 |
| Figure 6b: Pure titanium peg and sleeve system. |
 |
| Figure 6c: Hydroxyapatite-coated titanium sleeve with several titanium pegs. |
Thus, although pegs allow a more lifelike quality to the artificial eye, they are not for everyone.
b) Peg Variations (Figure 6a)
the original peg was made of plastic (polycarbonate). A hole was drilled into the implant and a standard peg was put in place. To obtain a more secure fit between the orbital implant and peg, a peg and sleeve system was designed. Following drilling of a hole into the implant, a sleeve is screwed into the implant until it is tight and flush with the implant surface. A peg is then placed into the center of the sleeve. In more recent years titanium has replaced polycarbonate as a peg and sleeve material since it is better tolerated in the socket tissue `(more biocompatible).
Some companies produce pure titanium peg systems (Figure 6b) whereas others produce hydroxyapatite-coated titanium (Figure 6c).
The hydroxyapatite coating results in significantly greater interface strength than uncoated titanium.
6) Socket Reconstructive Procedures
a) Volume Augmentation - some degree of sunkenness is common in artificial eye patients. The appropriate selection of an adequate implant size at the time of enucleation or evisceration is the first step in decreasing the sunken appearance of the artificial eye patient. However, if there remains some sunkenness, techniques are available to decrease it. One option is to undergo a second surgical procedure to implant a second implant (sled or floor implant) into the eye socket, underneath and behind the first. A general anaesthetic is required to put these volume augmentation implants into position. They are designed to slip in underneath and posterior to the first implant. They can be secured in position by glue, wire or a mini plate system used in facial reconstruction. The surgery is short (45 minutes) and patients are discharged with a patch in place on the day of surgery or the following morning. The artificial eye remains in position but may require an adjustment in the first few weeks. Pain is not a big factor as there is very little disruption to the socket tissues (Figure 7a,b).
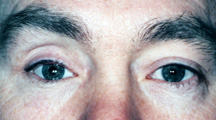 |
| Figure 7a: Sunken socket appearance. |
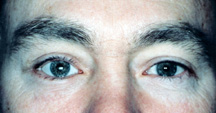 |
| Figure 7b: 3 months following an orbital floor implant - the sunken appearance is gone. |
Another technique to decrease a sunken appearance involves the use of “dermal fat” grafts. A graft of fat just beneath the skin (“dermal fat”) can be harvested from the hip of the patient. This fat is then implanted into the sunken appearing upper lid (sulcus). The hip incision lies beneath the underwear or bathing suit area. The fat is trimmed and implanted into the sunken appearing upper lid (sulcus) making a 1-inch long eyelid skin crease incision. A pocket is made for the fat graft, which is then implanted followed by skin closure. This procedure is routinely done under local or local stand-by anaesthesia (twilight anaesthesia) as a day patient. It is not painful and has good to excellent results. Over correction is required as some fat atrophy does occur in the first 3 months.
Lastly, to balance the sunken appearance of the artificial eye, removal of a small amount of skin and fat from the upper lid of the opposite seeing eye can be performed. This is a very simple and quick procedure that is routinely done as an outpatient. Sometimes it can be performed with nice results even prior to consideration of the “floor implants” or “fat graft technique” described.
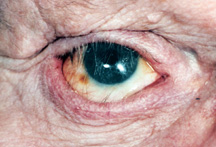 |
| Figure 8: With a shallow inferior fornix the lower lid appears pushed down and the lashes are rolled upward toward the prosthesis. |
b) Fornix Reconstruction - After many years of wearing an artificial eye, recurring socket infection and/or scarring secondary to trauma, the pocket behind the lower lid where the artificial eye sits (“inferior fornix”) can become shallow. This may be associated with a retracted appearing lower lid (a lower lid that looks too low) as well as artificial eye fitting problems with the artificial eye recurrently falling out (Figure 8).
To correct this, an initial assessment by the ocularist can be performed to determine if a modified custom made prosthesis might be of some benefit. If not, a fornix deepening surgical procedure is required. One such surgery involves borrowing some of the lining from the inside of the mouth (mucous membrane) and using it to create a deeper lower lid pocket. Hard palate mucosa (from the palate of the mouth) can also be used as well as ear cartilage or donor sclera. These surgeries are done under local stand-by anaesthesia (twilight anaesthesia) or general anaesthesia. They generally take 1-1½ hours and are very well tolerated. If hard palate mucosa is used, hot foods and liquids may be difficult to tolerate for a few weeks.
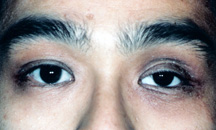 |
| Figure 9: Drooping of the left upper lid. |
7) Eyelid Reconstructive Surgeries
a) Ptosis of the upper lid - Ptosis refers to “drooping.” A droopy upper lid or "ptotic upper lid” is not uncommon. After several years of wearing an artificial eye, and removing it on numerous occasions, it is possible for the upper lid elevation muscle (levator aponeurosis) to become thin and stretch. As a result, the upper lid falls and becomes “ptotic” (Figure 9).
If the lid droop is mild, an adjustment to the artificial eye may elevate the lid and correct the droop. If the lid remains droopy, a simple, quick, painless operation can be performed referred to as “levator advancement.” The surgery takes approximately 15-30 minutes and is done under local freezing as an outpatient. Post-op there may be some minor bruising and swelling in the first week.
b) Lower lid laxity - After many years of wearing an artificial eye, the lower lid, (which supports the weight of the artificial eye) may become lax (loose). As a result the lower sags downward. To correct this, a minor lid tightening procedure can be performed. Under local anaesthesia as an outpatient, a snip is made in the lateral part of the lid and the firm part of the lid (tarsal plate) is reconnected to the bony orbital rim. This procedure, commonly referred to as a “tarsal strip” is simple to do. It takes about 15 minutes and relatively pain free. The sutures used are dissolvable and only rarely cause some tenderness while absorbing.
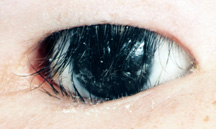 |
| Figure 10: Entropion of the upper and lower eyelids - notice the eyelashes resting on the surface of the prosthesis. |
c) Entropion repair - Entropion refers to an inward turning of the lid (Figure 10). In the artificial eye patient it may be seen on the upper and lower lid. As entropion occurs, the lashes become more vertical in the upper and lower lid, and may end up resting on the prosthetic eye. Surgery for upper or lower lid entropion is available, generally as an outpatient under local anaesthesia. The surgeries usually last 30 minutes and the goal is to rotate the lashes either in the upper or lower lid back to a more normal position. The sutures used are absorbable and dissolve over 3-6 weeks depending upon what type is used. Post-op there may be mild swelling and bruising. Pain is not a major concern but some minor discomfort may be present.
d) Ectropion repair - Ectropion refers to an outward turning of the lower lid. After several years of artificial eye wearing and tissue laxity development in the lower lid, the lower lid may not only sag downward but may, in some instances turn away from the artificial eye (become ectropic). Like entropion, this problem can be corrected by a very straightforward outpatient procedure under local anaesthetic with absorbable sutures. The goal is to return the eyelid to a more normal position so that it sits against the artificial eye. Surgery lasts 15-30 minutes and may be associated with some minor lid swelling and bruising.
8) Concluding Remarks About Artificial Eyes
Eye contact is an essential part of human interaction. It is extremely important for the artificial eye patient to maintain a natural, normal-appearing prosthetic eye. In recent years major developments have taken place in reconstruction of an eye socket following enucleation/evisceration or secondary implant surgery. The ideal orbital implant has been sought for more than a century. Porous materials (hydroxyapatite, porous polyethylene and aluminum oxide) are currently the preferred orbital implants primarily because of the vascularization and tissue integration that can occur. These implants are less likely to migrate than previously used plastic implant and are associated with a higher degree of motility especially when coupled to the overlying artificial eye through a peg system. Which implant is best is currently a matter of debate. The ideal porous implant is one that is biocompatible, bioinert, non-toxic, non-allergenic, inexpensive and stable over time.
References:
1. Labat B. Chanson A., Frey J. Effects of alumina and hydroxyapatite coatings in the growth and metabolism of human osteoblasts. J Biomed Mater Res 1995; 29:1397 - 1401
2. Mawn L, Jordan DR, Ahmad I , Gilberg S. Effects of orbital biomaterials on human fibroblasts. Can J Ophthalmol 2001; 36:245 - 251
3. Christel P. Biocompatibility of surgical grade dense polycrystalline alumina.Clin Orthop 1992; 282: 10 - 18